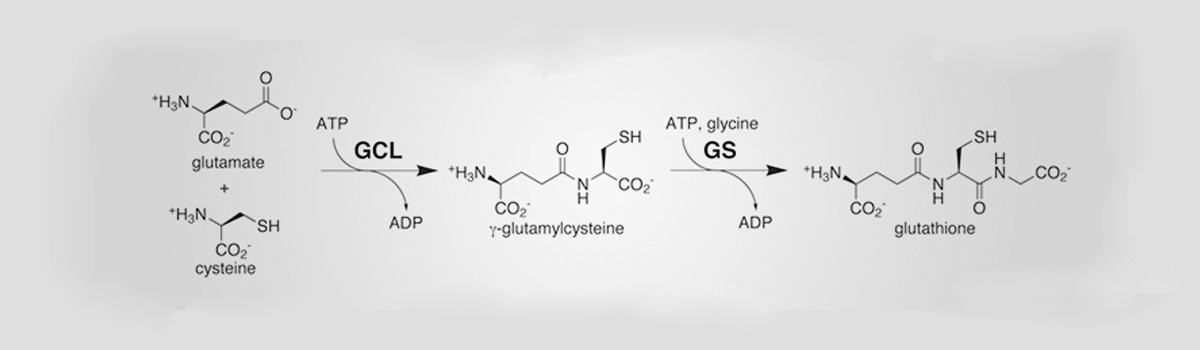
Glutathione is made up of three amino acids: Glutamate, Cysteine and Glycine. It is produced in every cell and is synthesized by two enzymes. The first enzyme, glutamate cysteine ligase (GCL), joins the amino acids glutamate and cysteine together to form gamma-glutamylcysteine (Glyteine). The second enzyme, glutathione synthase (GS), adds the amino acid glycine to Glyteine to form glutathione. Though Glyteine is produced continuously in every cell of the body, almost all of it is immediately converted to glutathione. The Glyteine concentration in cells at any given time is therefore negligible.