Unfortunately, there are many claims on how to enhance cellular glutathione effectively. One of the most common claims is that the amino acid cysteine is in limited supply in the body. We are aware that cysteine is one of the three building blocks that make up glutathione, but is there any evidence to suggest that we may be low on cysteine? And, regardless, would taking cysteine be effective in increasing cellular glutathione? On initial observation, the principle behind the theory of cysteine deficiency being a cause of low glutathione appears reasonably sound, but it is not that simple.
The first question is relatively easy to answer. The fact is that our diet usually contains plenty of cysteine and the other sulphur-containing amino acid methionine, which is readily converted into cysteine in the liver [1]. For example, the typical American diet supplies much more than the recommended required quantity of cysteine [2]. We can, therefore, rule out a cysteine deficiency. But would taking a cysteine or cysteine supplement such as N-acetylcysteine (NAC) increase our cellular glutathione?
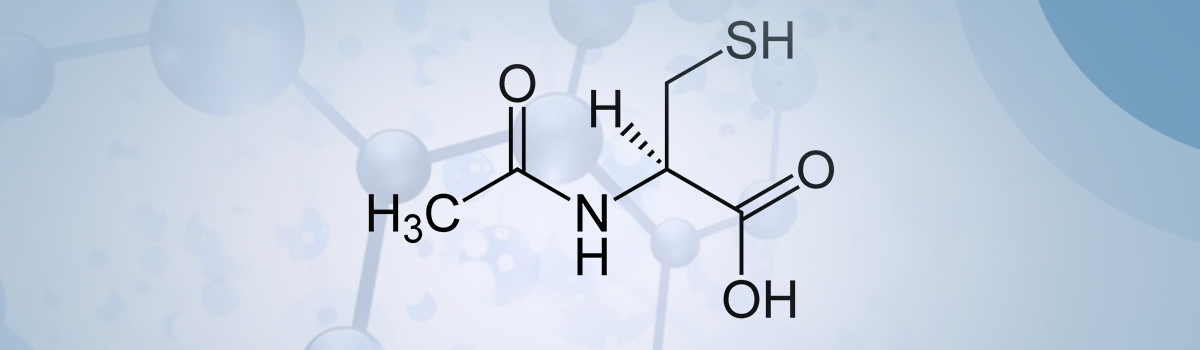
Cysteine, unlike most other amino acids, is extremely unstable and rapidly autoxidizes to cystine which is the oxidized disulphide form. It has exceedingly low solubility, and it is not absorbed from the GI tract. Additionally, this cysteine autoxidation reaction, catalyzed by transition metal ions, generates oxygen free radicals and hydrogen peroxide. In high concentrations, this may result in cellular toxicity [3-6] and has the potential to be neurotoxic [7]. Our cells have adapted to this potential toxicity by storing cysteine in the form of glutathione [8], which is far more stable to oxidation. We can hence regard glutathione to be a safe storage for cysteine. It is important to note that consuming cysteine as part of our usual diet will never exceed the threshold to become toxic.
There is a notable exception that relates to acute glutathione depletion due to acetaminophen (paracetamol) overdose, as we shall see.
By far, the most studied cysteine supplement is the cysteine prodrug NAC. Several human clinical studies have determined the bioavailability of NAC. Orally delivered NAC undergoes extensive first-pass metabolism resulting in about 90% loss by enzymic deacylation to form cysteine in the small intestine [9]. AAs we have seen, this mainly gets converted to cystine and is of little use in healthy individuals or those suffering from a chronic undersupply of glutathione due to aging or illness. One notable exception is the observation in significant studies that NAC is highly effective in elevating glutathione under conditions where there has been a dramatic (acute depletion) drop in intracellular glutathione levels, for instance, as is the case in acetaminophen overdose. Here, the sharp decline in glutathione levels, especially in the liver, to almost zero is effectively counteracted by NAC [10]. It immediately supplies cysteine for repletion of glutathione and thus, recovery from toxicity. Unfortunately, this is where NAC has gained a false reputation as a go-to drug if low glutathione is suspected. While immensely helpful indeed, it does not address the problem of supplementing glutathione in cases of gradual depletion such as chronic illness or just simply getting older.
In contrast, diseases in which there is a prolonged and chronic decrease in glutathione do not respond well to NAC treatment. An example of this is the situation that occurs in HIV/AIDS patients who experience a persistent drop in tissue glutathione levels. In a clinical trial of AIDS patients were treated with 1.8 g/day of NAC for two weeks with the glutathione status monitored in plasma and lymphocytes. During the treatment, no significant increase in glutathione was observed [11]. Similar disappointing observations of HIV patients supplemented with NAC were also made by [12] and [13]. NAC has been tried in numerous chronic diseases with similarly disappointing results, including cystic fibrosis protection against contrast-induced nephropathy and thrombosis [14].
The tight negative feedback control that glutathione exerts on the first of two enzymes responsible for glutathione synthesis, GCL, can explain this phenomenon. This enzyme has the task of combining the amino acids glutamate and cysteine to form Glyteine, which is eventually needed to produce glutathione by the next enzyme GS. As long as cellular glutathione is above a level considered to be adequate, which is called homeostasis, GCL is inhibited from making Glyteine, no matter how much cysteine is available. However, when intracellular glutathione is well below this homeostatic level, GCL is no longer inhibited and can actively utilize the cysteine that NAC supplies. Several researchers have come to the same conclusion when trying to explain this fact [15]. Negative feedback controls exist as part of many of our body’s processes to tightly regulate certain functions; for example, our body temperature.
Supplementing with cysteine to increase glutathione is therefore of little use, except in a few severe and limited cases mainly used in clinical settings. We now understand the causes of glutathione depletion during aging and chronic illness and how to effectively, rapidly and safely augment cellular glutathione with Glyteine.
In summary, taking a cysteine supplement is of little use to increase glutathione because our body tightly regulates both the storage and production of cysteine and any excess we consume is broken down into more stable by-products.
References